COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) Pipeline Report including Stages of Development, Segments, Region and Countries, Regulatory Path and Key Companies, 2022 Update
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) are used to amplify and detect the specific nucleic acid sequences of SARS-CoV-2 virus at the point-of-care.
The COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) pipeline market research report provides comprehensive information about the COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) pipeline products with comparative analysis of the products at various stages of development and information about the clinical trials which are in progress. Moreover, the report also provides information about various pipeline products and their estimated approval dates.
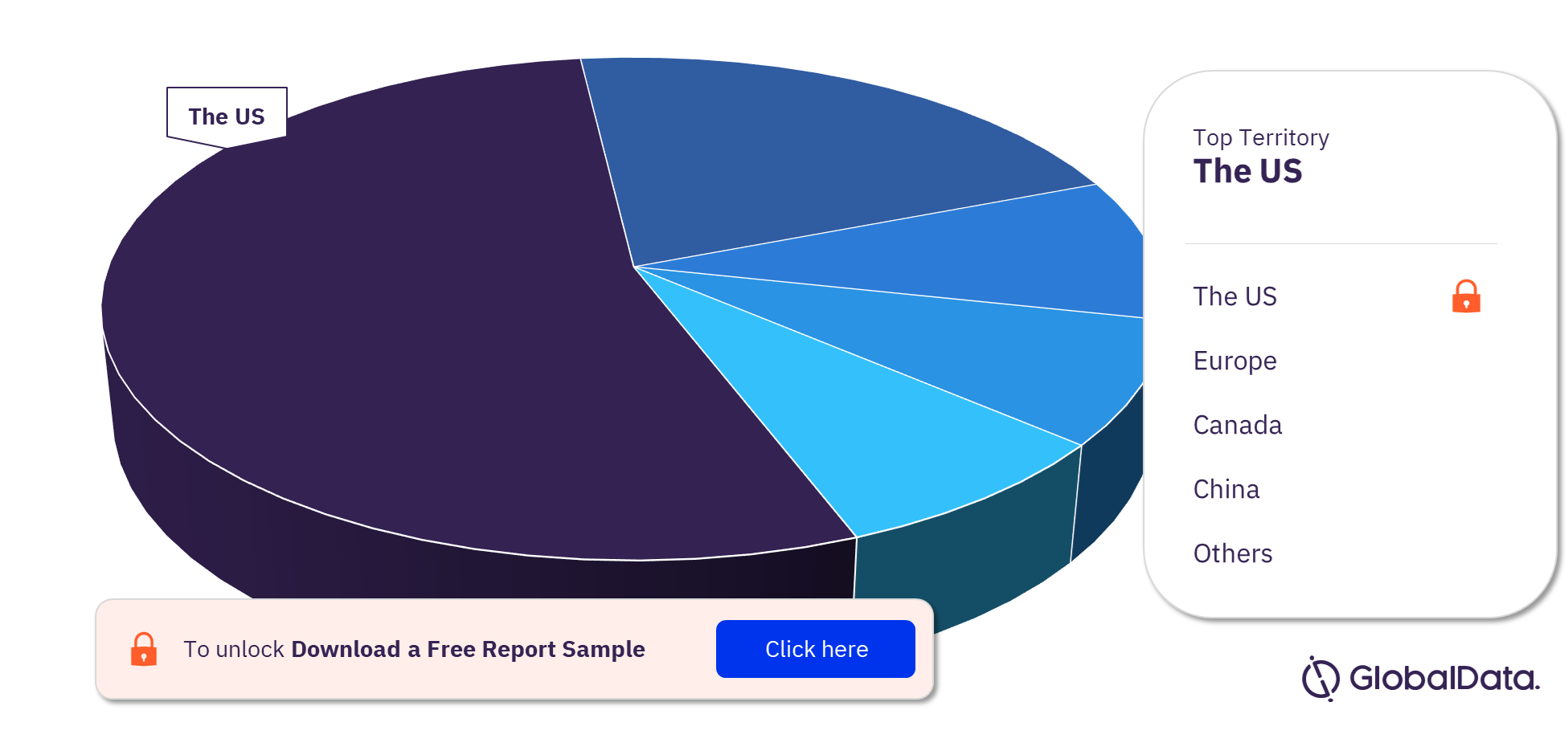
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Segmentation by Territories
Some of the key territories with products in the pipeline are the US, Europe, Canada, China, India, United Kingdom, Ireland, Japan, Kuwait, and Singapore. The US is the leading territory in the COVID 19 POC NAATs pipeline products market.
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Analysis by Territories, 2022 (%)
For more territory insights into the COVID 19 POC Nucleic Acid Amplification Tests (NAATs) pipeline products market, download a free report sample
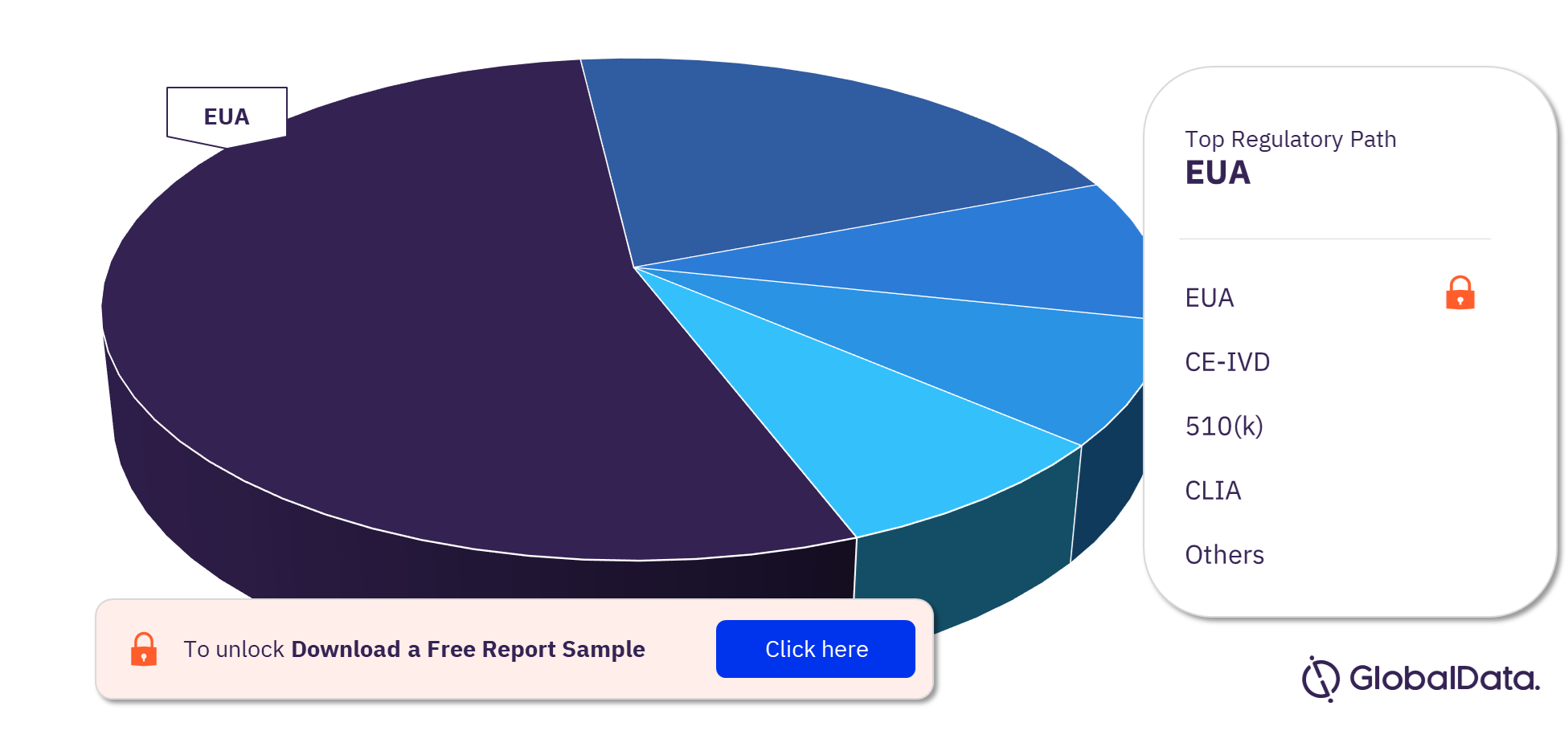
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Segmentation by Regulatory Paths
The COVID 19 POC Nucleic Acid Amplification Tests (NAATs) pipeline report provides detailed insights into the pipeline products by regulatory path including EUA, CE-IVD, 510(k), CLIA, de novo, MDL, NMPA, and PMA. Most of the products follow the EUA pathway to enter the market.
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Analysis by Regulatory Paths, 2022 (%)
For more regulatory path insights into the COVID 19 POC Nucleic Acid Amplification Tests pipeline products market, download a free report sample
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market - Competitive Landscape
Some of the leading companies in the COVID 19 Point-of-Care (POC) NAATs pipeline products market are Aegea Biotechnologies Inc, Aegis Sciences Corp, Alveo Technologies, Anavasi Diagnostics, Anitoa Systems LLC, Antigen Discovery Inc, Aptorum Group Ltd, Attoplex Co Ltd, Baebies Inc, and Bhushan J. Toley Lab.
Aegea Biotechnologies Inc: Aegea Biotechnologies Inc (Aegea) is a biotechnology company that discovers and develops reagents, research kits and in-vitro diagnostic (IVD) products for molecular analysis. The company is headquartered in San Diego, California, the US.
Aegis Sciences Corp: Aegis Sciences Corp (Aegis Sciences) operates and manages a forensic toxicology and healthcare laboratory that is engaged in offering science-driven drug testing and consulting services including urine and blood specimen testing, prenatal testing, and other testing services. The company is headquartered in Tennessee, the US.
Alveo Technologies: Alveo is a pre-clinical stage medical diagnostics company. The company is headquartered in Alameda, California, the US.
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Report Overview
| Key Territories | The US, Europe, Canada, China, India, United Kingdom, Ireland, Japan, Kuwait, and Singapore |
| Key Regulatory Paths | EUA, CE-IVD, 510(k), CLIA, de novo, MDL, NMPA, and PMA |
| Leading Companies | Aegea Biotechnologies Inc, Aegis Sciences Corp, Alveo Technologies, Anavasi Diagnostics, Anitoa Systems LLC, Antigen Discovery Inc, Aptorum Group Ltd, Attoplex Co Ltd, Baebies Inc, and Bhushan J. Toley Lab |
Segments Covered in the Report
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Territories Outlook
- The US
- Europe
- Canada
- China
- India
- United Kingdom
- Ireland
- Japan
- Kuwait
- Singapore
COVID 19 POC Nucleic Acid Amplification Tests Pipeline Products Market Regulatory Paths Outlook
- EUA
- CE-IVD
- 510(k)
- CLIA
- de novo
- MDL
- NMPA
- PMA
Scope
- Extensive coverage of the COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) under development
- The report reviews details of major pipeline products which includes, product description, licensing and collaboration details and other developmental activities
- The report reviews the major players involved in the development of COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) and list all their pipeline projects
- The coverage of pipeline products based on various stages of development ranging from Early Development to Approved / Issued stage
- The report provides key clinical trial data of ongoing trials specific to pipeline products
- Recent developments in the segment / industry
Reasons to Buy
The report enables you to:
- Formulate significant competitor information, analysis, and insights to improve R&D strategies
- Identify emerging players with potentially strong product portfolio and create effective counter strategies to gain competitive advantage
- Identify and understand important and diverse types of COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) under development
- Develop market-entry and market expansion strategies
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline
- In-depth analysis of the product’s current stage of development, territory and estimated launch date
Aegis Sciences Corp
Alveo Technologies
Anavasi Diagnostics
Anitoa Systems LLC
Antigen Discovery Inc
Aptorum Group Ltd
Attoplex Co Ltd
Baebies Inc
Bhushan J. Toley Lab
Biocartis Group NV
BioLayer Corporation Limited
BiologyWorks Inc
Biyani Biosolutions Pvt Ltd
CAMPTON Diagnostics UG
CardiAI Inc
Columbia University
Columbia University Fertility Center
Coyote Bioscience Co Ltd
Credo Diagnostics Biomedical Pte Ltd
Cue Health Inc
Curiosity Diagnostics Sp zoo
DiaCarta Inc
DNA Xperts Pvt Ltd
Eurofins Scientific SE
FemtoDx Inc
Flambeau Diagnostics
Fluxergy LLC
genedrive plc
Hangzhou FasTech Biotechnology Co Ltd
Harvard University
ID Genomics Inc
Jiangsu Qitian Gene Biotechnology Co Ltd
KogeneBiotech Co Ltd
Lamprogen Inc
Lucira Health Inc
Luminex Corp
Lynntech Inc
Mammoth Biosciences Inc
McMaster University
Meridian Bioscience Inc
Merlin Biomedical (Xiamen) Co Ltd
Mesa Biotech Inc
Microgem International Plc
Midge Medica GmbH
NDFOS Co Ltd
OxDX Ltd
Patanjali Pharma Pvt Ltd
PathogenDx, Inc.
Pepex Biomedical, Inc.
Prenetics Ltd
Prime Discoveries Inc
Princeton University
Prominex Inc
QuikPath Pte Ltd
RNA Disease Diagnostics Inc
Sense Biodetection Ltd
Sentebiolab Biotechnology
Shenzhen Second People's Hospital
Shimadzu Corp
Shionogi & Co Ltd
Tangen Biosciences Inc
Todos Medical
Uh-OH Labs Inc
University of California Berkeley
University of Illinois at Urbana-Champaign
University of Texas Southwestern Medical Center
Veredus Laboratories Pte Ltd
Visby Medical Inc
XP Diagnostics GmbH
Table of Contents
Table
Figures
Frequently asked questions
-
Which are the key territories in the COVID 19 POC Nucleic Acid Amplification Tests pipeline products market?
Some of the key territories in the COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) pipeline products market are the US, Europe, Canada, China, India, United Kingdom, Ireland, Japan, Kuwait, and Singapore.
-
What are the key regulatory paths in the COVID 19 POC Nucleic Acid Amplification Tests pipeline products market?
The key regulatory paths in the COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) pipeline products market are EUA, CE-IVD, 510(k), CLIA, de novo, MDL, NMPA, and PMA.
-
What are COVID 19 POC Nucleic Acid Amplification Tests used for?
The COVID 19 Point-of-Care (POC) Nucleic Acid Amplification Tests (NAATs) are used to amplify and detect the specific nucleic acid sequences of SARS-CoV-2 virus at the point-of-care.
-
Which are the leading companies in the COVID 19 POC Nucleic Acid Amplification Tests pipeline products market?
Some of the leading companies in the COVID 19 POC Nucleic Acid Amplification Tests (NAATs) pipeline products market are Aegea Biotechnologies Inc, Aegis Sciences Corp, Alveo Technologies, Anavasi Diagnostics, Anitoa Systems LLC, Antigen Discovery Inc, Aptorum Group Ltd, Attoplex Co Ltd, Baebies Inc, and Bhushan J. Toley Lab among others.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more In Vitro Diagnostics reports